Oscillometry Healthcare Professional Toolkit
What is respiratory oscillometry? Definition, clinical applications and interpretation overview for healthcare professionals.This toolkit is designed to help healthcare professionals better understand respiratory oscillometry, also referred to as forced oscillation technique (FOT). Impulse oscillometry (IOS) is a sub-technique of respiratory oscillometry. You can review the sections below on clinical perspectives and mechanics, as well as various tools and attachments. For more details on oscillometry and citations, please refer to the downloadable toolkit. While this toolkit does not go into the details of oscillometry interpretation, it does provide a brief overview.
Oscillometry Basics
Respiratory oscillometry is an objective lung function test that provides different information about the respiratory system that complements traditional lung function tests like spirometry and body plethysmography. Oscillometry is a non-invasive method for assessing the respiratory mechanics of the lung tissue (elasticity/stiffness), airways, and chest wall (compliance) during normal tidal breathing.
An oscillometry device generates sound waves, which are superimposed on a patient's normal tidal breathing, to measure respiratory resistance and reactance (elastance or stiffness).
This effort-independent lung function test is different from spirometry. Oscillometry requires:
- Passive breathing only – no forced maneuvers
- Minimal cooperation or coordination from the patient
Therefore, it is useful for young children, those who cannot perform acceptable spirometry maneuvers and, in many situations, where spirometry is contraindicated.
Oscillometry Overview
Clinical Perspectives
Oscillometry applies different frequencies of airflow or pressure, through sound waves, at the mouth during normal breathing. These sound waves are usually taken within a frequency range of 5 to 40 Hertz, which is higher than the frequency of normal breathing. The device then measures the oscillating pressure or flow generated by the lungs in response.
Oscillometers used different techniques to generate these waves. The forced oscillation technique (FOT) delivers multi-frequency sinusoidal waves whereas the impulse oscillometry systems (IOS) deliver square waves as trains of individual frequencies.
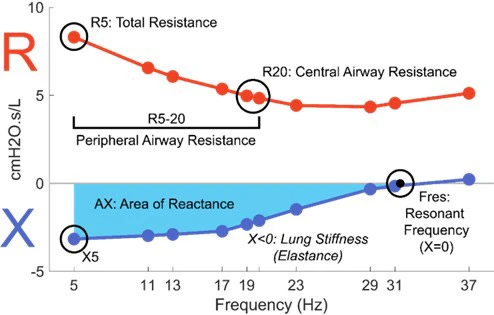
Oscillometry measures impedance (total opposition to airflow), which has components:
- Resistance (R): The degree of airway obstruction in the lungs (aka openness of airways)
- Reactance (X): The compliance (stiffness/elastance) of the lung tissue (aka expandability of airways)
Impedance = Resistance + Reactance
- Impedance: is the total opposition to airflow
- Resistance (R): The degree of airway obstruction in the lungs
- Reactance (X): The compliance (stiffness/elastance) of the lung tissue. How much the lungs can stretch and recoil.
Oscillometry, like spirometry and plethysmography, are objective lung function tests. This chart provides a comparison between these two objective lung function tests.
Spirometry and Oscillometry: Evidence Based Comparison Chart
| Feature | Spirometry | Oscillometry |
|---|---|---|
| What this objective test measures best | Large airway dynamics | Peripheral / small airway dynamics |
| Key measures | Measures lung volumes and airflow:
| Measures respiratory impedance using small amplitude oscillatory multi-frequency waves delivered at the mouth to measure respiratory:
|
| Primary use | Gold-standard for the diagnosis of airflow obstruction (asthma, COPD) | Highly sensitive to small-airway dysfunction, therefore useful across asthma, COPD, and other conditions for early detection or response to treatment |
| Level of patient effort required | Requires forceful, maximal effort, often difficult for young children, elderly, or patient with illness | Effort-independent, requiring only tidal breathing – easier for young children and elderly and can be used in situations where spirometry is contra-indicated |
| Sensitivity to small-airway disease | Limited; FEF 25-75 may correlate but inconsistently | Superior sensitivity to small airway dysfunction, early detection of COPD symptoms and airway abnormalities |
| Measured dynamically or at rest | Challenges airways through forced expiratory flow which reveals abnormalities | Measures airways during normal breathing conditions and identifies abnormalities at rest |
| Bronchodilator response detection | Changes in FEV1/FVC used to assess responsiveness | Changes in Resistance (R) and Reactance (X) used to assess responsiveness More sensitive to detect bronchodilator responsiveness |
| Test duration | Longer – requires repeated coached maneuvers and rest periods | Shorter, quick to perform (30–60 second maneuvers) |
| Key metric to aid in medication therapy | FEV1 guides medication therapy in asthma – NHLBI 2007 FEV1, along with patient’s symptoms and exacerbation risk, guide COPD therapy | No established metric to guide medication therapy |
| Patient burden | May provoke cough, breathlessness, and fatigue (e.g. IPF) | Associated with less symptom burden |
| Ideal populations | Older children and adults who can follow instructions | Anyone but particularly very young children (>3 years), patients with cognitive or language barriers through lifespan |
The Mechanics of Oscillometry
In oscillometry, sound waves are artificially generated pressure oscillations that ride on top of a person’s normal breathing (tidal breathing).
1. A loudspeaker (or a piston or a vibrating mesh) inside the oscillometry device is used to generate sound waves.
- The speaker diaphragm moves back and forth.
- This motion creates small pressure fluctuations in the air—basically sound waves.
2. How the oscillations enter the lungs
- The patient breathes normally through a mouthpiece.
- While the patient breathes, the device superimposes these sound waves into the airflow.
- The oscillations are low-amplitude (gentle) and usually low frequency (about 5–35 Hz).
3. As the sound waves travel inside of the airways
- Some energy is resisted by airway narrowing and friction.
- Some energy is stored and released by the elastic tissues of the lungs.
- Some energy reflects back due to changes in airway size.
This interaction changes the phase and amplitude of the sound waves.
4. Sensitive sensors measure:
- Pressure changes
- Flow changes
From these pressure and flow changes, the oscillometry device calculates respiratory impedance (total opposition), which includes:
- Resistance (R) (airway obstruction)
- Reactance (X) (stiffness/elastance)
5. Sound waves are perfect to be used for lung function measurement because they:
- Travel easily through air
- Can probe different airway sizes depending on frequency
- Do not require forced breathing
By measuring how these waves change as they travel through the airways, the device can calculate respiratory impedance (total opposition), separating airway resistance (airway obstruction) and reactance (stiffness/elastance). Using different frequencies helps the system assess both large and small airways.
The following factors should be considered when selecting an oscillometer for a clinic setting:
- FDA approval for intended clinical use
- Clinical validation data demonstrating accuracy, repeatability, and sensitivity
- Measurement parameters available (e.g., R5, R20, X5, AX, Fres)
- Quality control algorithms (coherence, leak detection, artifact rejection)
- Reference equations included and appropriateness for patient population
- Age range and feasibility (pediatric, adult, elderly)
- Ease of operation and consistency between operators
- Patient coaching aids (real‑time feedback, animations, visual cues)
- Turnaround time per test for busy clinic workflows
- Report clarity to aid clinical interpretability
- Data connectivity (electronic health record integration, export formats)
- Physical footprint and portability within clinic spaces
- Consumables and infection control compatibility
- Calibration procedures and ongoing maintenance needs
- Total cost of ownership (device, software, disposables, service)
- Vendor support, software updates
- Training availability
Oscillometry is a reimbursable lung function test.
CPT Code for Oscillometry
- 94728: Airway resistance by impulse oscillometry.
Coding considerations
- Bundling: Code 94728 cannot be reported with certain other pulmonary function tests, such as spirometry (94010) or bronchodilator response (94060). If billed with another CPT code with a status indicator of S, T or V, it will be bundled (i.e. 94010, 94060, 94070, 94375, 94726).
- Specificity: Always check the specific type of test performed to ensure the correct code is used, as there may be other codes for different types of airway resistance or lung volume measurements.
- Modifiers: Modifiers may be needed in certain situations. For example, Modifier 26 (Professional Component used when billing only the provider’s professional work, but not the equipment, supplies, or technical side) and TC (Technical Component when the provider bills only the technical portion of a service and not the provider’s interpretation) can be used when the professional and technical components of the service are provided by different entities.
Page last updated: March 24, 2026